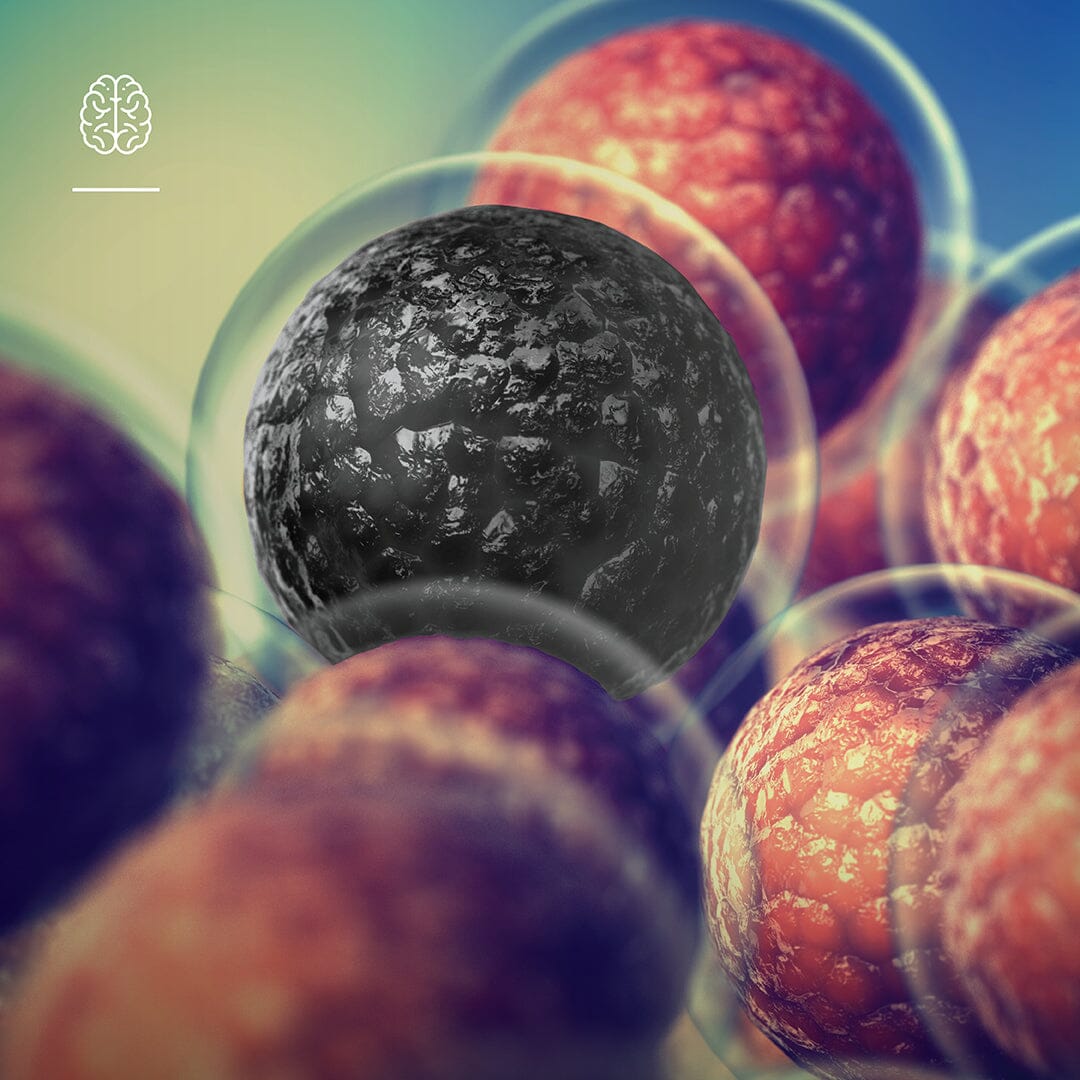
The term “zombie cells” brings to mind images of the living dead… and correctly so. It’s a great analogy to describe what happens to cells that are no longer of use to us, entering a zombie like state. The scientific term is senescent cells and recent science is focussed on how we can reduce the burden of these cells in your body as we age.
Cellular Senescence
Senescent cells are cells that have reached the end of their productive life. These cells enter a catatonic like state which gives the appearance that they are neither dead nor alive.
Senescent cells pose little problem in our youth and instead represent an important cellular process that protects your body from cellular abnormalities. All being well, your body can identify abnormal cells or tissues, such as scar tissue that has completed its task, and stop them from proliferating and leading to damage and disease4.
How does the body get rid of senescent cells?
Redundant or “zombified” cells secrete inflammatory molecules into the blood stream. These molecules act as a signal to the immune system, alerting immune cells to initiate the removal of these cells from our circulation4.
The removal of senescent cells is facilitated by a process known as autophagy. This process supports healthy cells by clearing away the damaged cell components. These components are then recycled to create new cells and so the cycle continues.
During this process, the damaged cell components are consumed by cellular structures known as a lysosomes – if you imagine Pac Man, lysosomes act in a similar way, engulfing the degraded material in a protective membrane. Lysosomes then digest its contents using enzymes, which breaks down the components into molecules. These molecules are released outside the cell to be circulated in the blood stream and recycled according to the needs of your body.
Autophagy is not limited to the removal of degraded cellular debris from senescent cells. Broadly, autophagy is the process whereby unwanted or unrequired cellular components are removed – for example, bacteria, viruses or cancerous cells.
There are three types of autophagy – chaperone mediated autophagy, micro-autophagy and macro-autophagy. These categorise the different autophagy mechanisms available to transport unwanted cellular components into the lysosome¹.
When do senescent cells become problematic?
As we age (and especially in overweight individuals), our immune systems become less responsive to these signals or are less efficient at the clean-up operation. This is when senescent cells can become problematic to the body and over time lead to or exacerbate many age-related diseases.
When the body becomes less efficient at clearing senescent cells, they accumulate in the body. These cells continue to secrete inflammatory molecules and this leads to toxicity and increasing inflammation in the body – this phenomenon is often referred to as “inflammaging”².
The good news is that emerging science has identified cellular senescence and autophagy as mechanisms that can be supported by therapeutic interventions.³ ⁴
Fisetin supports the healthy levels of senescent cells and autophagy
Fisetin is a natural compound, known as a flavonoid, found in a variety of fruits and vegetables such as strawberries, apples and persimmons. It assists in targeting and removing senescent cells which are resistant to programmed cell death.
In a 2018 study on mouse and human tissue, ten flavonoids were tested for their senolytic potential, or in other words, the ability of these molecules to support the clearance senescent cells from the body. Fisetin was found to have the strongest effect of the ten flavonoids in the removal of senescent cells.⁵
Intermittent fasting to promote autophagy
Over the past decade numerous studies have been published on different forms of fasting or calorie restriction to stimulate autophagy and promote wellbeing and longevity.
A review of over 30 studies found that:
“Both fasting and calorie restriction have a role in the upregulation of autophagy, the evidence overwhelmingly suggesting that autophagy is induced in a wide variety of tissues and organs in response to food deprivation”⁶.
Suzy Walsh
BBA (Hons)., BNat., mNMHNZ
Registered Naturopath & Medical Herbalist
[1] Parzych, K. R., & Klionsky, D. J. (2014). An overview of autophagy: Morphology, mechanism, and regulation. Antioxidants & Redox Signaling, 20(3), 460-473. doi: 10.1089/ars.2013.5371
[2] Camell, C. (2021). Senolytics reduce coronavirus-related mortality in old mice. Innovation in Aging, 5(Supplement_1), 247-248. doi: 10.1093/geroni/igab046.951
[3] Robbins, P. D., et al. (2021). Senolytic drugs: Reducing senescent cell viability to extend health span. Annual Review of Pharmacology and Toxicology, 61(1), 779-803. doi: 10.1146/annurev-pharmtox-050120-105018
[4] V. Gorgoulis, et al. (2019). Cellular Senescence: Defining a Path Forward. Cell, 179, 813-829. doi:10.1016/j.cell.2019.10.005 Medline
[5] Yousefzadeh, M. J., et al. (2018). Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine, 36, 18-28. doi:10.1016/j.ebiom.2018.09.015
[6] Bagherniya, M., Butler, A. E., Barreto, G. E., & Sahebkar, A. (2018). The effect of fasting or calorie restriction on autophagy induction: A review of the literature. Ageing Research Reviews, 47, 183-19. doi: 10.1016/j.arr.2018.08.004

![Never Grow Old - Longevity Issue [Nutrition Business Journal]](http://scienceresearchwellness.com/cdn/shop/articles/NBJ_Post_-_LinkedIn.jpg?v=1692742979&width=1500)
![Circulatory system care "vital" [NZHERALD]](http://scienceresearchwellness.com/cdn/shop/articles/SRW_-_News_Clip_-_Post_-_Cir1_-_D1_dfadc7dc-c57a-4f80-82bd-4fdb5ba5f0cd.jpg?v=1692742734&width=1500)
